Global Pharmaceutical Regulatory, Clinical & Pharmacovigilance Compliance Consulting Services
Your Compliance, Our Mission
Supporting pharmaceutical, herbal, and nutraceutical companies with seamless global regulatory compliance. Our experienced team assists from dossier development to product registration, validation, PV services, and DMF preparation—ensuring faster, compliant market entry.
Precision Takes Time, Quality Has Value
If you want work done right, at the right price, on the right timeline—you’re in the right place.
Industries We Serve
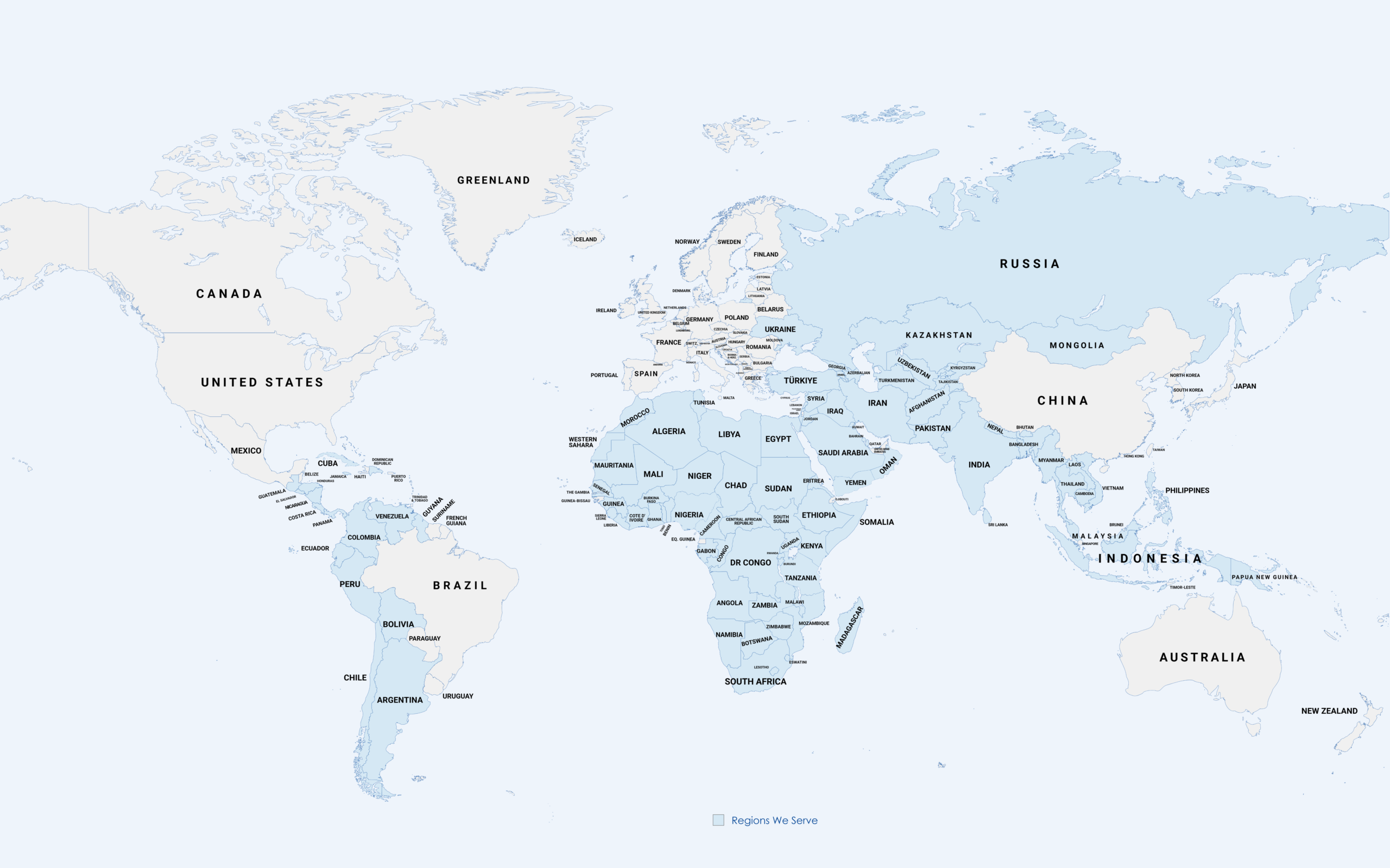
Supporting pharmaceutical companies across manufacturing and regulatory sectors in ASEAN, Latin, CIS, African, and other emerging markets.
Our Pharmaceutical Regulatory Services
End-to-end regulatory, clinical, pharmacovigilance, and compliance solutions for pharmaceutical companies worldwide.
Regulatory Intelligence
We have developed our advanced Regulatory Intelligence tools to streamline processes, monitor global regulatory changes, improve document quality, and deliver faster, more accurate compliance solutions.

Why Choose Regcure Pharma?

Our Track Record
Global Regulatory Expertise
Regcure Pharma supports pharmaceutical regulatory submissions
across ASEAN, Latin America, CIS, Africa, and other emerging markets.
Our Mission & Vision
Need Support with Pharmaceutical Regulatory Submissions?
Partner with Regcure Pharma to simplify regulatory compliance and accelerate your global market access.
