A bioequivalence (BE) study is a critical component in the development and regulatory approval of generic pharmaceutical products. Its purpose is to scientifically demonstrate that a generic drug performs in the same way as its reference (innovator) product in terms of safety, efficacy, and pharmacokinetic behavior. Achieving this requires precise planning, strict regulatory compliance, and accurate scientific execution.
Behind every successful BE study is a highly coordinated multidisciplinary team of professionals working across clinical, analytical, statistical, and regulatory domains. From the sponsor who initiates the study to the investigators, clinical staff, bioanalytical scientists, statisticians, and quality assurance professionals who ensure compliance and data integrity, each expert plays a crucial role in the success of the trial.
Because bioequivalence studies involve human volunteers, complex analytical techniques, and strict regulatory oversight, even minor errors in study conduct, sample handling, or data analysis can compromise the entire study. Therefore, the expertise and coordination of the bioequivalence study team are essential to ensure reliable results, ethical conduct, and regulatory acceptance.
In this article, we explore the key experts involved in a BE study and explain how their roles contribute to generating scientifically sound and regulatory-compliant outcomes.
Why a Multidisciplinary Team is Essential in a Bioequivalence Study
Bioequivalence studies operate at the intersection of clinical research, pharmaceutical science, analytical chemistry, biostatistics, and regulatory compliance. Because of this complexity, no single professional can manage all aspects of the study. Instead, a coordinated team of specialists works together to ensure that every stage of the study is conducted accurately, ethically, and in accordance with global regulatory standards.
During the clinical phase, medical professionals such as principal investigators, physicians, nurses, and clinical research coordinators oversee volunteer screening, dosing procedures, safety monitoring, and blood sample collection. Their work ensures that study subjects are protected and that clinical procedures follow Good Clinical Practice (GCP) guidelines.
In the bioanalytical phase, laboratory scientists and analytical chemists quantify drug concentrations in biological samples using highly sensitive techniques such as LC-MS/MS. Accurate analytical data are critical because they form the foundation for pharmacokinetic calculations that determine whether the test product is bioequivalent to the reference product.
The biostatistical and pharmacokinetic teams then analyze the data to calculate parameters such as Cmax, Tmax, and AUC. Using statistical models, they evaluate whether the test and reference products meet regulatory bioequivalence limits, typically within the accepted 80–125% confidence interval.
Equally important are the quality assurance, regulatory, and data management teams, who ensure that the study complies with Good Laboratory Practice (GLP), Good Clinical Practice (GCP), and regulatory requirements from authorities such as the FDA, EMA, and CDSCO. They monitor documentation, audit study procedures, and maintain data integrity so that the results withstand regulatory inspection.
Because bioequivalence studies demand precision, ethical oversight, and strict regulatory compliance, collaboration among these experts is essential. Each member of the bioequivalence study team contributes specialized expertise that collectively ensures the study produces reliable, scientifically valid, and regulatorily acceptable results.
Key Personnel in a BE Study & Their Responsibilities

1. Sponsor
- Provides study funding and overall support.
- Selects and authorizes the Contract Research Organization (CRO).
- Reviews and approves the study protocol.
- Submits regulatory documents and oversees study conduct.
2. Principal Investigator (PI)
- Acts as the overall in-charge of the study.
- Ensures protocol adherence and volunteer safety.
- Reviews Case Report Forms (CRFs) and verifies study data.
- Signs off on the final study report.
3. Sub-Investigator / Co-Investigator
- Supports the Principal Investigator in study execution.
- Conducts medical assessments and monitors adverse events.
- Ensures adherence to study protocol and GCP standards.
4. Clinical Research Coordinator (CRC)
- Manages subject scheduling and trial logistics.
- Maintains essential trial logs and documentation.
- Assists the PI in operational aspects of the study.
5. Clinical Research Associate (CRA)
- Monitors study sites and ensures compliance with GCP and SOPs.
- Verifies accuracy of collected data and source documents.
- Conducts site audits and ensures regulatory readiness.
6. Ethics Committee (EC/IRB)
- Reviews and approves the study protocol and informed consent forms.
- Ensures ethical conduct and protection of participant rights and safety.
- Oversees adherence to ethical guidelines throughout the study.
7. Regulatory Authority (e.g., CDSCO, FDA, EMA)
- Reviews and approves BE study applications.
- Conducts inspections of CRO facilities.
- Evaluates final reports for marketing authorization.
8. Study Physician / Medical Officer
- Conducts subject screening and medical examinations.
- Evaluates clinical laboratory data and monitors adverse reactions.
- Provides medical oversight during dosing and post-dose observation.
9. Pharmacist / Drug Dispenser
- Receives, stores, and dispenses test and reference drug products.
- Maintains investigational product accountability records.
- Ensures proper storage conditions and expiry tracking.
10. Clinical Laboratory Personnel
- Conducts safety laboratory tests such as CBC, LFT, KFT, and urinalysis.
- Maintains analytical precision and laboratory quality control.
11. Phlebotomist
- Collects blood samples at predefined time points.
- Ensures correct sample labeling, handling, and quality preservation.
12. Nurses / Clinical Staff
- Monitor vital signs and ECGs before and after dosing.
- Supervise drug administration and manage emergency responses.
- Provide continuous subject care during confinement.
13. Volunteer / Study Subject
- Participates voluntarily and follows study rules.
- Provides informed consent and cooperates during procedures.
- Undergoes sampling, monitoring, and medical assessments.
14. Data Manager
- Manages electronic data capture systems and databases.
- Ensures accurate data entry, validation, and discrepancy resolution.
- Oversees data cleaning and database locking before statistical analysis.
15. Biostatistician
- Designs the statistical model for the BE study.
- Performs ANOVA and calculates pharmacokinetic parameters (Cmax, AUC, Tmax).
- Determines bioequivalence within the regulatory confidence interval (80–125%).
16. Pharmacokinetic Scientist (PK Scientist)
- Interprets pharmacokinetic data and calculates PK parameters.
- Prepares the PK report for inclusion in the final study dossier.
17. Bioanalytical Scientist
- Develops and validates analytical methods (e.g., LC-MS/MS).
- Conducts analysis of plasma samples and ensures assay accuracy.
- Maintains method reproducibility as per FDA/EMA validation guidelines.
18. Quality Assurance (QA) Manager
- Ensures compliance with GCP, GLP, and SOPs.
- Conducts audits of clinical and bioanalytical activities.
- Reviews documentation and maintains audit trails for regulatory inspections.
19. Project Manager
- Coordinates all study teams and tracks project timelines.
- Manages communication between the sponsor and CRO.
- Oversees deliverables and ensures milestones are achieved.
20. Archivist / Documentation Specialist
- Manages secure storage of raw data, CRFs, and consent forms.
- Maintains the Trial Master File (TMF) for future reference.
- Ensures document traceability and archival integrity.
21. Contract Research Organization (CRO)
- Executes the BE study on behalf of the sponsor.
- Handles clinical, analytical, biostatistical, and reporting activities.
- Ensures overall compliance with regulatory standards.
22. Safety Officer / Pharmacovigilance Personnel
- Monitors adverse events (AE) and serious adverse events (SAE).
- Manages emergency medical responses and follow-ups.
- Ensures all safety events are documented and reported to regulators.
23. Food & Diet Supervisor
- Prepares standardized meals for study volunteers.
- Ensures diet compliance to avoid food-drug interactions affecting PK outcomes.
24. Housekeeping & Maintenance Staff
- Maintains cleanliness of clinical and sample storage areas.
- Provides sanitation support during study operations.
- Assists in maintaining controlled environments during subject confinement.
25. Security Staff
- Controls entry and exit of study subjects and staff.
- Maintains integrity of study protocols and prevents unauthorized access.
- Supports facility security during confinement and study operations.
26. IT / Data Security Personnel
- Ensures secure data storage and system backup.
- Manages hardware, software, and network security.
- Safeguards data integrity and confidentiality across all digital platforms.
Clinical Phase Roles
- Screening of subjects
- Informed consent process
- Drug administration
- Vital signs monitoring
- Blood sample collection
- Subject safety tracking
Bioanalytical Phase Roles
- Sample processing & storage at specified temperatures (deep freezer −20°C / −80°C)
- Sample run & validation as per USFDA/EMA guideline.
- Chromatogram review & documentation.
Biostatistical Phase Roles
- ANOVA analysis
- 90% CI calculation
- Test/Reference ratio assessment (80%-125% limits)
Documentation and QA Roles
- Audit trail
- QC checks
- SOP compliance
- Archival & retention
Summary Flow
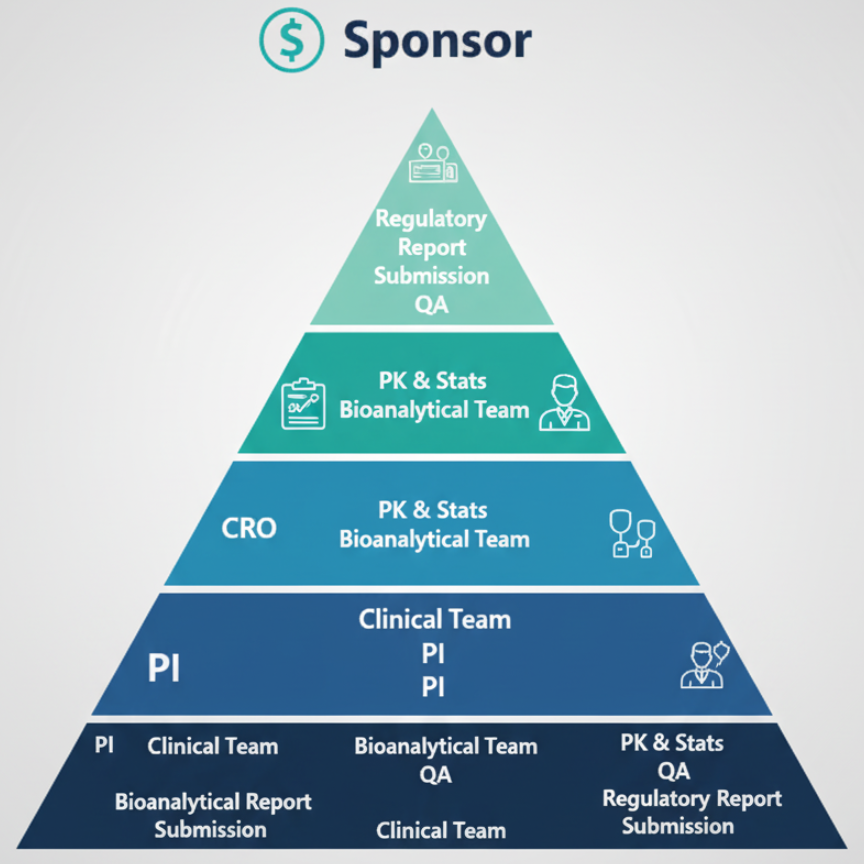
Bonus: Responsibilities Highlight
- PI = Study head & subject safety
- Sponsor = Funding & regulatory submission
- Bioanalytical Scientist = Sample analysis accuracy
- Statistician = BE conclusion validity
- QA = Study quality & audit
📩 Reach out to Regcure Pharma For expert regulatory support, clinical research insights, and end-to-end healthcare solutions: 📧 info@regcurepharma.com | 🌐 www.regcurepharma.com | 📞87990 45524





