NAFDAC dossier requirements 2025 introduce stricter regulatory standards for pharmaceutical product registration in Nigeria. Companies planning submissions must align with updated bioequivalence guidelines, submission portal rules, and pharmacovigilance obligations.
1. Understanding NAFDAC Dossier Requirements 2025
Over the past decade, NAFDAC has strengthened its regulatory framework to ensure that only safe, effective, and high-quality products reach Nigerian consumers. As Nigeria’s pharmaceutical market continues to expand, the agency’s focus has shifted toward:
- Harmonization with global standards such as the International Council for Harmonisation (ICH).
- Improved review efficiency through structured submission timelines.
- Enhanced product safety monitoring via ongoing pharmacovigilance measures like the Periodic Safety Update Report (PSUR).
These changes are not merely procedural; they signal a move toward a risk-based, science-driven evaluation system. Companies that adapt quickly will gain a competitive advantage, while those who delay may face extended approval times, rejection of applications, or even cancellation of ongoing reviews.
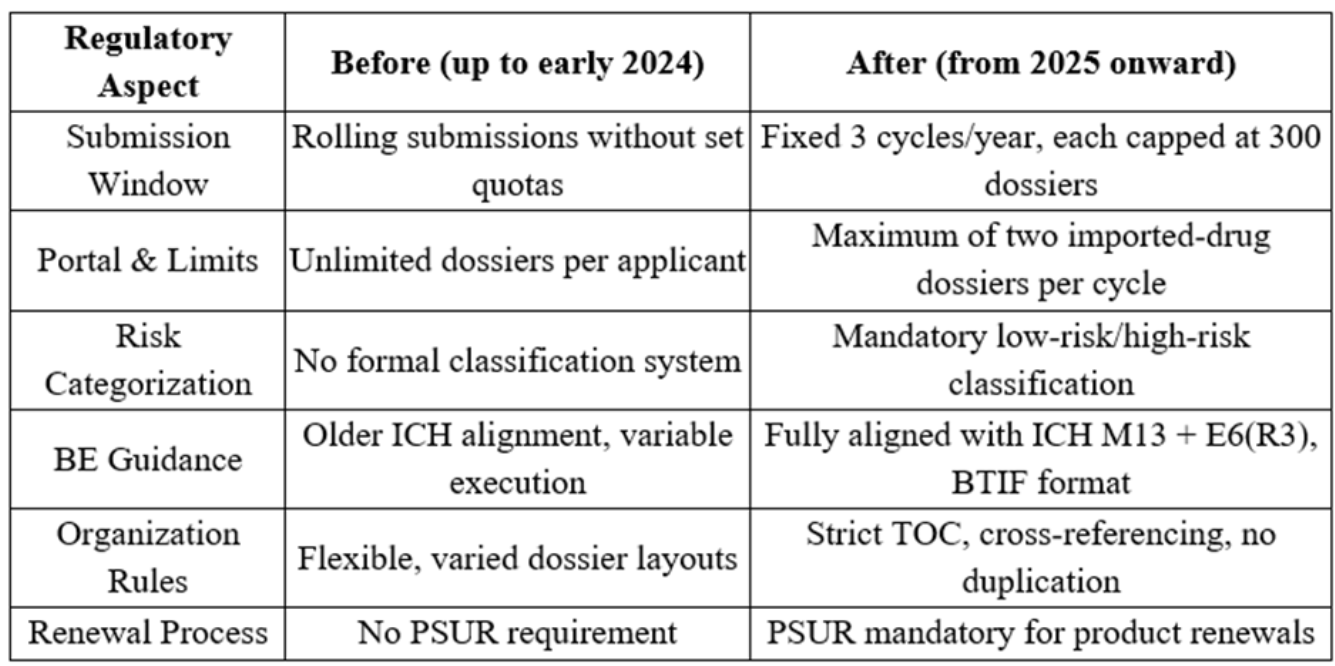
By examining the table above, it becomes clear that NAFDAC is moving from a flexible but inconsistent approach to a structured, capacity-controlled, and evidence-based model.
2. Submission Windows and Portal Rules — A New Era of Structured Timelines
On 29 April 2024, NAFDAC’s Dossier Management System (DMS) Version 2 reintroduced fixed submission windows and portal restrictions, reshaping how applicants plan their filings.
Key Changes
- Three Submission Cycles/Year – Max 300 dossiers per cycle.
- Two Imported-Drug Limit – Per applicant, per cycle.
- Strict File Formats – QIS/QOS in Word; BMRs in PDF. Incorrect formats = auto rejection.
Why It Matters Missing a cycle could delay launches by months due to limited slots.
Best Practices
- Plan at least six months ahead.
- Audit documents for correct format/naming.
- Keep DMS account updated and tested.
- Use reliable, high-speed internet for uploads.
By adapting to these rules, applicants can secure a submission slot and reduce regulatory delays.
3. Risk-Based Categorization — A Smarter Approach to Dossier Evaluation
Since late 2024, all human drug dossiers must be classified as:
- Low-Risk – e.g., oral solid generics, low-absorption topicals.
- High-Risk – e.g., sterile injectables, biologics, controlled substances.
Benefits: Speeds up low-risk reviews while ensuring deep scrutiny for high-risk products.
Risks of Misclassification: Delays, rejections, and additional study requirements.
Best Practices:
- Follow NAFDAC’s latest guidelines.
- Align with WHO/ICH standards.
- Document your classification rationale.
Regcure Support: Expert classification, tailored documentation, and compliance checks.
4. Updated Bioequivalence (BE) Guidelines — Effective May 2025
From May 2025, NAFDAC will enforce revised Bioequivalence (BE) requirements aligned with ICH M13 and ICH E6(R3), ensuring Nigerian medicines meet international quality, safety, and efficacy standards.
Key Updates
- Study Design: Standard single-dose crossover; parallel for long half-life drugs.
- Sampling Duration: Minimum 72 hours.
- Meal Composition: Defined for fed-state studies.
- Washout Period: Standardized and justified.
- Protocol Format: Mandatory BTIF.
- Compliance: Full GCP/GLP adherence.
- Statistics & Data: Detailed plan, full traceability, electronic validation.
Implications for Applicants
- Correct design selection (crossover vs. parallel).
- Logistics for extended sampling.
- Standardized nutrition control.
- Robust statistical evaluation.
- Strict BTIF formatting and documentation.
Challenges Higher costs, protocol rejections, operational complexity, and audit risks.
Best Practices
- Start protocol planning 6–9 months before submission.
- Partner with CROs experienced in ICH M13/E6(R3).
- Conduct pre-submission audits.
- Use secure electronic data capture.
- Train all staff on GCP/GLP compliance.
How Regcure Pharma Helps
- BE protocol design per NAFDAC & ICH standards.
- Clinical trial oversight and data accuracy.
- BTIF preparation and submission.
- Audit readiness for NAFDAC inspections.
5. Stricter Clarity and Completeness Requirements
NAFDAC now enforces:
- Defined TOC — Follow official structure.
- No Duplication — One upload per document, cross-reference instead.
- Precise Cross-Referencing — Include section and page numbers.
- File Naming Rules — Match NAFDAC’s format exactly.
Even strong dossiers can be rejected for poor organization.
Regcure Support: Structural audits, correct document placement, file format compliance.
6. PSUR Requirement for Renewals — Strengthening Post-Marketing Surveillance
NAFDAC now mandates Periodic Safety Update Reports (PSURs) for marketing authorization renewals, aligning with ICH E2C(R2) and global pharmacovigilance standards to ensure medicines remain safe and effective post-approval.
PSUR Overview
- Comprehensive benefit–risk assessment after approval.
- Includes cumulative safety data, adverse event analysis, and global regulatory status updates.
Why It Matters?
- Shifts focus to post-market safety in renewal decisions, protecting patients and ensuring compliance.
Submission Scope
- Required each renewal cycle; interim updates for high-risk products.
- Covers data since last PSUR or initial approval.
- Must follow ICH E2C(R2) format.
Key Challenges
- Global data collection.
- Tight timelines.
- Aligning NAFDAC and global standards.
Best Practices
- Maintain ongoing pharmacovigilance.
- Use standardized templates.
- Engage cross-functional teams.
- Include international safety developments.
- Perform internal quality checks.
Regcure Pharma Support We handle complete PSUR preparation, integrate global data, align with NAFDAC timelines, and deliver expert benefit–risk analyses for smooth renewals.
7. Challenges & Pitfalls for Applicants — Navigating the New Regulatory Landscape
Frequent Issues:
- Incomplete dossiers.
- Misclassification of product risk.
- Non-compliance with formats.
- Missing BE or PSUR data.
- Last-minute submissions.
- Use of unqualified dossier preparers.
Avoid By:
- Preparing months ahead.
- Following NAFDAC-specific formats.
- Keeping documents updated.
- Conducting pre-submission audits.
Regcure Support: Dossier review, category guidance, compliance checks, strategic planning.
8. Regcure Pharma’s Strategic Support — Your Partner in NAFDAC Compliance & Market Success
🚀 Navigating NAFDAC’s Evolving Regulatory Landscape — The Regcure Pharma Advantage
In Nigeria’s fast-changing pharmaceutical regulatory environment, success depends on more than just meeting current NAFDAC requirements — it’s about anticipating changes, avoiding compliance gaps, and responding quickly to queries.
At Regcure Pharma, we deliver end-to-end regulatory solutions tailored for the Nigerian market:
- ✅ Dossier Preparation & Review – Audit-ready, NAFDAC-compliant submissions for faster approvals.
- ✅ Risk-Based Product Categorization – Accurate classification to avoid reclassification delays.
- ✅ Bioequivalence (BE) Coordination – CRO selection & compliant BE reports to prevent post-submission surprises.
- ✅ PSUR & Renewal Support – Timely submissions to maintain uninterrupted market presence.
- ✅ Regulatory Query Management – Evidence-based responses to speed up approvals.
- ✅ Strategic Regulatory Planning – Aligning launches with NAFDAC cycles for optimal time-to-market.
Why Choose Us?
- Deep Nigerian regulatory expertise with a global perspective.
- Preventive strategies to reduce costly queries.
- Transparent communication & progress tracking.
With Regcure Pharma, you get more than a submission service — you gain a compliance partner committed to your market success.
📞 Let’s ensure your next NAFDAC submission is fast, compliant, and stress-free.





